 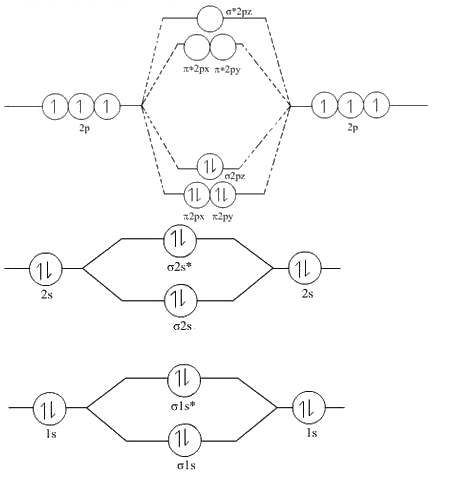
A molecular orbital describes the behavior of one electron in the electric field generated by the nuclei and some average distribution of the other electrons. Most present-day methods in computational chemistry begin by calculating the MOs of the system. They are invaluable in providing a simple model of bonding in molecules, understood through molecular orbital theory. Most commonly a MO is represented as a linear combination of atomic orbitals (the LCAO-MO method), especially in qualitative or very approximate usage. A molecular orbital can specify the electron configuration of a molecule: the spatial distribution and energy of one (or one pair of) electron(s). Instead they are obtained from the combination of atomic orbitals, which predict the location of an electron in an atom. However calculating the orbitals directly from this equation is far too intractable a problem. Molecular orbitals are approximate solutions to the Schrödinger equation for the electrons in the electric field of the molecule's atomic nuclei. Molecular orbitals are of three types: bonding orbitals which have an energy lower than the energy of the atomic orbitals which formed them, and thus promote the chemical bonds which hold the molecule together antibonding orbitals which have an energy higher than the energy of their constituent atomic orbitals, and so oppose the bonding of the molecule, and non-bonding orbitals which have the same energy as their constituent atomic orbitals and thus have no effect on the bonding of the molecule.Ī molecular orbital (MO) can be used to represent the regions in a molecule where an electron occupying that orbital is likely to be found. They can be quantitatively calculated using the Hartree–Fock or self-consistent field (SCF) methods. They are usually constructed by combining atomic orbitals or hybrid orbitals from each atom of the molecule, or other molecular orbitals from groups of atoms. Mathematically, molecular orbitals are an approximate solution to the Schrödinger equation for the electrons in the field of the molecule's atomic nuclei. The electrons from the constituent atoms occupy the molecular orbitals. When multiple atoms combine chemically into a molecule by forming a valence chemical bond, the electrons' locations are determined by the molecule as a whole, so the atomic orbitals combine to form molecular orbitals. In an isolated atom, the orbital electrons' location is determined by functions called atomic orbitals. At an elementary level, they are used to describe the region of space in which a function has a significant amplitude. Mulliken in 1932 to mean one-electron orbital wave functions. The terms atomic orbital and molecular orbital were introduced by Robert S. This function can be used to calculate chemical and physical properties such as the probability of finding an electron in any specific region. In chemistry, a molecular orbital ( / ɒr b ə d l/) is a mathematical function describing the location and wave-like behavior of an electron in a molecule. The right column shows virtual MO's which are empty in the ground state, but may be occupied in excited states. The orbital wave functions are positive in the red regions and negative in the blue. The white and grey line visible in some MO's is the molecular axis passing through the nuclei. The left column shows MO's which are occupied in the ground state, with the lowest-energy orbital at the top. See also: Molecular orbital theory and Molecular orbital diagram Complete acetylene (H–C≡C–H) molecular orbital set.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
